You could almost feel the ground shift. Years from now, we might look back at January 2024 as an important pivot point in our understanding, classification, and approach to treatment of Parkinson’s disease (as we currently call it). Blink and you might have missed it, but a series of three articles and an associated editorial just appeared in the highly esteemed British medical journal The Lancet last week. Opting to run with a powerful quote rather than an image on the front cover (see below), it is fair to say the editors of The Lancet, a leading source of clinical and health knowledge since 1823, have understood the scope of the problem that is Parkinson’s disease and elected to highlight the urgent need for more research.

The editorial, titled “What next in Parkinson’s disease?”, calls attention to the fact that Parkinson’s disease, which is second only to Alzheimer’s disease among neurodegenerative disorders, is rapidly increasing in prevalence (estimated to hit 12–17 million people by 2040), adding to the already considerable strain on global health systems. Pointing to the need for more research and clinical studies, the editors state very clearly that “[a] disease-modifying treatment to slow, halt, and reverse progression of Parkinson’s disease must be the goal to strive for.”
The related series of three papers in the January 20 issue separately summarize the current state of knowledge on the epidemiology (population-level research) of Parkinson’s, our understanding of its causes (the underlying biology), and optimal treatments. These articles, authored by thought leaders in each of these three subfields, highlight the multiple factors contributing to Parkinson’s, from causative genes to risk-contributing genes, to pollutants and environmental toxins, to age-related changes, and how emerging treatments aim to address these underlying causes in a much more targeted and personalized manner. The article on treatments also devotes considerable space to the existing treatments for non-motor symptoms, which are often cited as more concerning than the motor symptoms by people living with Parkinson’s disease; this information will no doubt be highly informative for clinicians (i.e., the readers of The Lancet), leading to better disease management.
It is highly likely that past clinical trials of a range of promising treatments, while not successful in modifying the disease course in a broad group of patients, did indeed lead to benefit in some patients; reclassifying patients based on underlying causes and contributing factors (see below for more on this) therefore represents an opportunity to better design and power clinical trials for success. Watch this space!
Meanwhile, also last week, the February 2024 issue of The Lancet’s sister journal, The Lancet Neurology, was published online. This issue includes two biology-based frameworks for diagnosing and classifying (into subgroups) people with Parkinson’s disease (or in the case of one of the frameworks, the closely related “neuronal α-synuclein disease” or NSD). This is a significant paradigm shift that aims to enable earlier diagnosis and treatment (and testing of new treatments in clinical trials), overcoming the existing problem that a person’s Parkinson’s disease is often already quite advanced by the time of diagnosis using clinical criteria. Indeed, the disease can wreak havoc on important biological processes over many years before the clinical manifestation of “parkinsonism”, at which stage Parkinson’s disease is generally diagnosed. By then, well over half of an individual’s dopamine-containing neurons in the part of the brain called the substantia nigra—neurons that play a critical role in voluntary movement—have already been lost.
The Position Paper presenting the NSD concept, titled “A biological definition of neuronal α-synuclein disease: towards an integrated staging system for research”, also includes a staging system based on the presence or absence of certain features (see image below). Importantly, these stages extend proximally to include individuals at risk, those with genetic factors known to cause Parkinson’s (stage 0), and those with α-synuclein (the protein that misfolds and clumps together in Parkinson’s) pathology and dopamine dysfunction (stages 1A and 1B)—all of which precede the appearance of clinical signs or symptoms (stage 2) where disease would traditionally be diagnosed (if not later). While there are still many issues to be resolved, this work represents a big step toward earlier, accurate diagnosis of disease with the possibility of more targeted (biology-based), personalized treatment, and we applaud our friends at Cure Parkinson’s, Parkinson’s UK, Shake it Up Foundation Australia, and The Michael J Fox Foundation for Parkinson’s Research, among others, for supporting this work and playing an active role in its genesis. As the authors of the Position Paper state: “A biological definition of neuronal α-synuclein disease and an NSD-ISS research framework are essential to enable interventional trials at early disease stages. The NSD-ISS will evolve to include the incorporation of data-driven definitions of stage-specific functional anchors and additional biomarkers as they emerge and are validated.”
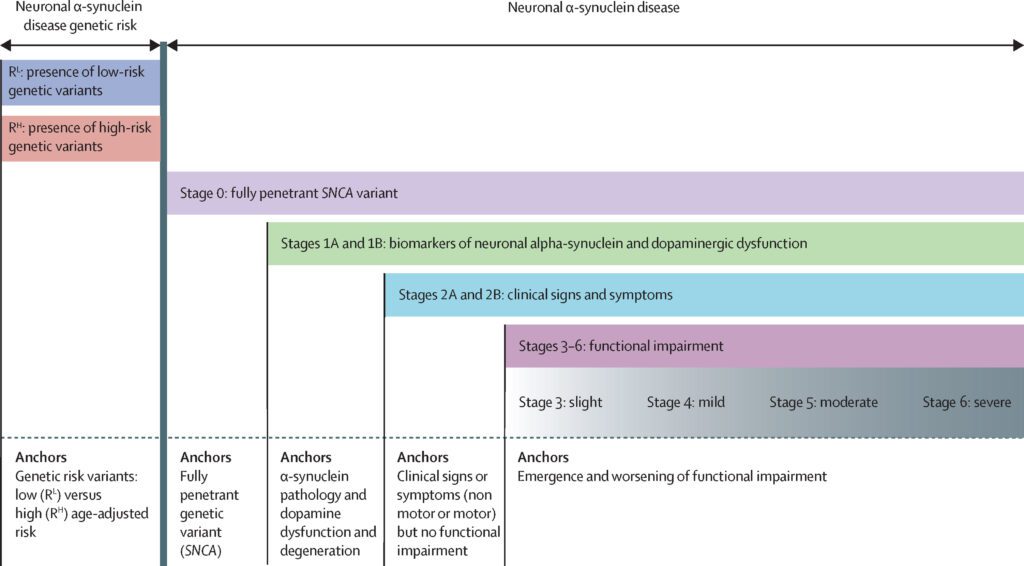
Figure: The NSD-ISS staging system. Reproduced from Simuni, Tanya, Lana M. Chahine, Kathleen Poston, Michael Brumm, Teresa Buracchio, Michelle Campbell, Sohini Chowdhury et al. “A biological definition of neuronal α-synuclein disease: towards an integrated staging system for research.” The Lancet Neurology 23, no. 2 (2024): 178-190.
The eagle-eyed among you will have noted that Parkinson’s disease is not explicitly mentioned in the title… this is deliberate. Parkinson’s disease and related disorders like dementia with Lewy bodies are currently defined based on their clinical features. The paradigm shift here is to define disease based on underlying biology; hence the name neuronal α-synuclein disease, which is not a clinically recognised condition. Indeed, the staging system is not intended to be used in a clinical setting (yet), but rather, as a tool for research to enable more effective testing of new treatments. However, there is likely to be a very high degree of overlap (>90%) between NSD and Parkinson’s, and the staging system will very much help tease apart the various subgroups sitting under the umbrella of Parkinson’s disease and closely related disorders.
The second biological framework, presented in a Personal View paper by a separate group of authors, is known as the SynNeurGe research diagnostic criteria. There are many points in common between the two frameworks, as highlighted by this helpful Comment article in the same issue, but the SynNeurGe criteria are not accompanied by a staging system like the NSD-ISS. The SynNeurGe criteria are based on the presence or absence of pathological α-synuclein in tissues or CSF, evidence of underlying neurodegeneration, and gene variants that cause or strongly predispose to Parkinson’s disease.
Both of these frameworks build on last year’s discovery of a biomarker for Parkinson’s (the ability to detect α-synuclein pathology in a sample from living individuals, which was highly predictive of Parkinson’s and even those deemed at risk but showing no clinical signs). There certainly remain some controversies and unanswered questions to be resolved, as clearly pointed out in the three Comment articles in the same issue; however, as the toolkit continues to grow and new tests are validated, there will be ever more opportunities to test potential treatments for their ability to modify the disease course and ultimately stop or slow progression of the second most common but fastest growing neurodegenerative disease.
Note on the cited publications
While The Lancet and The Lancet Neurology are subscription journals requiring payment for content, the articles highlighted here are all available free of charge. Please note that registration on the journals’ websites is required to be able to access these articles.


